Published: 18 Apr 2017
Category:
If you want to be an elite athlete, you have to pay attention to every detail. During the last Olympics in Rio, the Team GB medal haul was their best since 1908. Many commentators, including Sir Steve Redgrave attributed this incredible achievement to a new mind-set in UK sport called the ‘aggregation of marginal gains’. Simply put, this means breaking down the components that contribute to successful sporting performance and making every single one just a little bit better in a systematic, iterative way. Former British number one table tennis star Matthew Syed has written about the marginal gains concept in his 2015 book ‘Black Box Thinking: The Surprising Truth About Success’. We can apply this concept to refining techniques in animal research. Refinement requires the systematic analysis of the animal’s lifetime experience into its component steps, to identify potential sources of physical and psychological suffering and to put in place measures to eliminate or ameliorate this. In this article, we focus on the ‘R’ of refinement and present examples of how small changes to everyday procedures can be implemented to refine experimental procedures.
Refining blood sampling in mice
If you need to take a blood sample from a laboratory animal the first port of call in looking for refined procedures should be the excellent NC3Rs website, which provides species specific
information and guidance on method selection.
How much blood do you need?
The Home Office mandates that the maximum volume that can be taken is <10% total blood volume (TBV) on any single occasion to avoid hypovolaemic shock. For repeated blood samples this is typically limited to <15% TBV in 28 days. On average, mice have around 58.5 ml of blood per kg of bodyweight. So for an adult 30 g mouse TBV=1.75 ml and the maximum blood sample you can take is 175 μl. However, the biochemical methods used for analysing blood samples typically require small blood volumes. Smaller blood volumes (20-40 μl) should be taken in the case of repeated sampling or when using juvenile mice, which may weigh only 10-15g. So we should collect just the blood that we need.
What are your options?
There are a large number of methods of collecting a blood sample from a mouse. It’s important to think about your sampling techniques and how to ensure you only take what you need. For example, consider the blood volume that collects in the syringe: can you reduce the dead space? Also, if you plan to do behavioural pharmacology, the use of anaesthesia or invasive indwelling cannulae might confound the behaviour you are studying. A number of microsampling techniques and devices are available to limit blood volume collected.
Sadler and Bailey validated a refined method for repeated blood sampling in juvenile and adult mice using the tail incision method to sample from the lateral tail vein. The lateral tail vein is an appropriate route for repeated sampling of small blood volumes from mice without anaesthesia, although vasodilatation may be required which is in itself a potential stressor.
For this method the mouse is gently cupped by one person while the experimenter makes a small incision, about 2 cm from the tip of the tail, in the lateral tail vein using a razor blade (Figure 1). Blood is directly collected into small volume (20-40 μl as required) capillary tubes. Blood flow starts and stops spontaneously through the small incision or with a small amount of pressure so no blood is “wasted”. When corticosterone levels were used to assess whether repeated measurements, up to 3 times at 24h intervals, were stressful, there was no difference between corticosterone levels in samples taken on the third day compared with baseline.
Figure 1
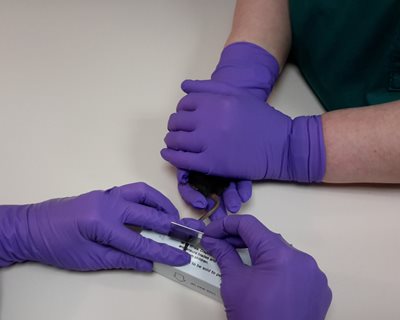
With this methodology Sadler and Bailey demonstrated a minimally invasive procedure for blood sampling that refined handling of the animals (no restraint devices were used), no anaesthesia was used, the smallest blood volume necessary was taken (and none wasted in needles or syringes), no tail warming was required and the procedure was demonstrated to be as stress-free as possible.
Additionally, the ability to take samples from the same mouse at multiple time points reduces the number of animals required for an experiment, compared to taking terminal blood samples from large numbers of mice. Thus, this method is in keeping with the principles of both refinement and reduction of animal use.
Refining restraint methods for drug dosing
For most pharmacological studies the experiment will involve the administration of substances to the animal by injection or via oral gavage. The details of how animals were handled during the administration of the substance is not normally reported in the literature beyond general statements such as ‘lightly restrained’. There are in fact a wide variety of methods used to restrain rodents for drug dosing. The most refined methods currently recommended are described on the
Procedures With Care website which includes video demonstrations of the techniques they recommend. These approaches all use some form of physical restraint which can be both stressful and aversive to the animal. Stress has the potential to have major effects on the results, changing an animal’s physiology and potentially altering the pharmacological response.
Restraint stress is one of the most common methods used to induce a negative affective state in animals and yet is used routinely as part of dosing procedures. It is also likely that the experience of the handler will have a major impact on the stress of the animal during dosing which can result in variability in the data and potentially limit reproducibility. Interestingly, methods to improve physical restraint have been adopted in other areas outside rodent research. For example, studies have shown that primates can be trained to accept injections and blood sampling without physical restraint and through the use of
positive reinforcement training. This approach has also been adopted in zoos where animals are trained to accept procedures through training, reducing the need for sedation, anaesthesia and the stress of restraint. So could the methods used to handle rodents be improved to minimise restraint so that stress is reduced and the processes refined?
A new approach
In 2015, Stuart and Robinson published a study reporting a modified handling technique for intraperitoneal dosing in rats which reduced measures of stress without impacting on the pharmacokinetics of the drug. The authors developed a method whereby animals were minimally restrained during intraperitoneal dosing using a cupping technique (see Figure 2). Rather than the conventional method of a firm restraint of the animal or scruff, the animal is positioned so that its abdomen is in a relaxed position. The animals show minimal response to the insertion of the needle in stark contrast to animals which have been restrained using a conventional method (
see online movie). The study found a ~50% reduction in corticosterone levels in animals dosed using this refined method as well as reduced behavioural measures of stress (reduced struggling, vocalisation and faecal count). The method was tested across a number of different rat strains and age groups with similar benefits observed. The approach was particularly beneficial in well-handled animals illustrating the added value of habituation of animals to procedures. Although not necessarily a technique which everyone will feel confident to adopt, the method clearly illustrates the advantages of reduced physical restraint during routine dosing methods. This is an important area for refinement as it can not only benefit the welfare of the animals but may also improve scientific outcomes. The work also highlighted the level of stress which even short restraint periods can trigger on an animal and the potential for this to adversely affect the results.
Figure 2
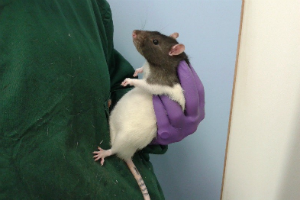
Hopefully these examples will encourage you to think about your experimental design and practice. Things like: methods of restraint, choice of vehicle, route of administration, changes to housing and care, use of peri- and post-operative analgesia, use of dedicated welfare score sheets and humane endpoints. It’s reassuring that when you talk to scientists many of these things are considered normal and “just how we do things” but they represent important refinements that can make a big difference to animal welfare. Perhaps we can all do more to report the refinements we use every day – big or small – and share good practice with the wider scientific community.
Comments
If you are a British Pharmacological Society member, please
sign in to post comments.